All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know GvHD.
The GvHD Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the GvHD Hub cannot guarantee the accuracy of translated content. The GvHD Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The GvHD Hub is an independent medical education platform, sponsored by Medac and supported through grants from Sanofi and Therakos. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View GvHD content recommended for you
The protective effect of leukemia inhibitory factor against GvHD after allo-BMT
Allogeneic bone marrow transplant (allo-BMT) is commonly used as a curative-intent therapy in patients with hematologic malignancies.1 However, development of graft-versus-host disease (GvHD) can limit the effectiveness and use of allo-BMT. Gastrointestinal tract involvement in GvHD can lead to increased GvHD severity and mortality, due to invasion of the intestinal stem cell compartment by donor T-cells, causing damage.1
Leukemia inhibitory factor (LIF) has been shown to be involved in regulating inflammation and maintenance of intestinal stem cell function and number.1 The mechanism by which LIF can provide a protective benefit against GvHD is shown in Figure 1. Here, we discuss a preclinical study by Wang et al.1 that investigated the role of LIF in GvHD using murine models.
Figure 1. LIF mechanism of action and its protective role in GvHD*
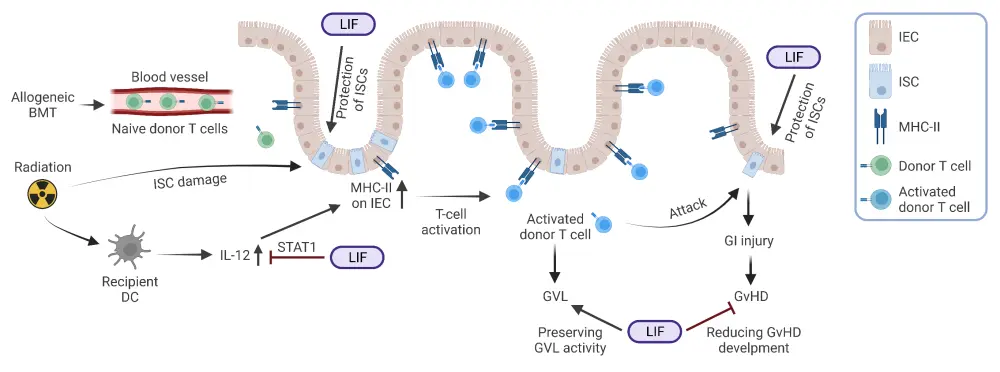
BMT, bone marrow transplant; DC, dendritic cell; GvHD, graft-versus-host disease; GVL, graft-versus-leukemia; IL, interleukin; IEC, intestinal epithelial cell; ISC, intestinal stem cell; LIF, leukemia inhibitory factor; MHC, major histocompatibility complex.
*Adapted from Wang, et al.1 Created with BioRender.com.
Study design
The mice used in this study were obtained from the Jackson Laboratory. The mice lines used and the BMT design are shown in Figure 2. Conventional LIF-knockout mice were also sourced from the European Mouse Mutant Archive (EMMA) repository.
Figure 2. Study design*
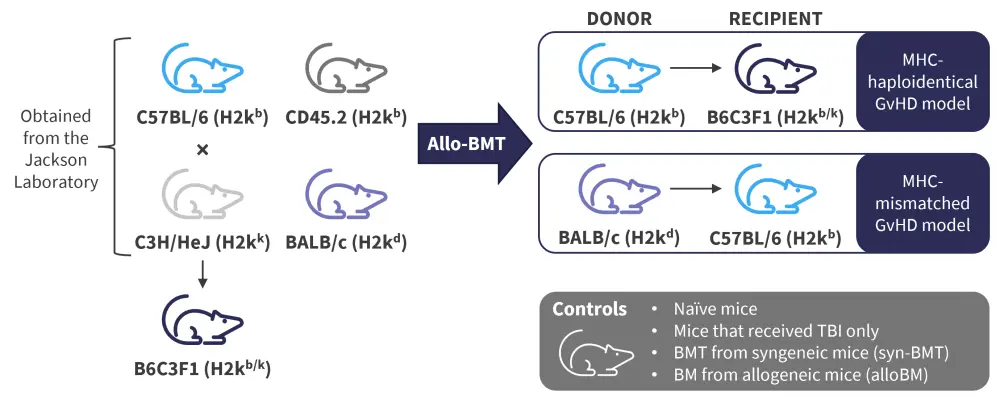
alloBM, allogeneic bone marrow; allo-BMT, allogeneic bone marrow transplant; BMT, bone marrow transplant; GvHD, graft-versus-host disease; MHC, major histocompatibility complex; syn-BMT, syngeneic bone marrow transplantation; TBI, total body irradiation.
*Data from Wang, et al.1
Mice used in this study were matched based on age (8−12 weeks of age) and sex. Transplant was given intravenously at a dose of 5 × 106 bone marrow cells. Outcomes measured in this study included serum LIF levels after BMT, development of GvHD, and the effect of administering recombinant LIF protein (rLIF). Figure 3 shows the irradiation, transplant, and rLIF schedule.
Figure 3. Transplant schedule*
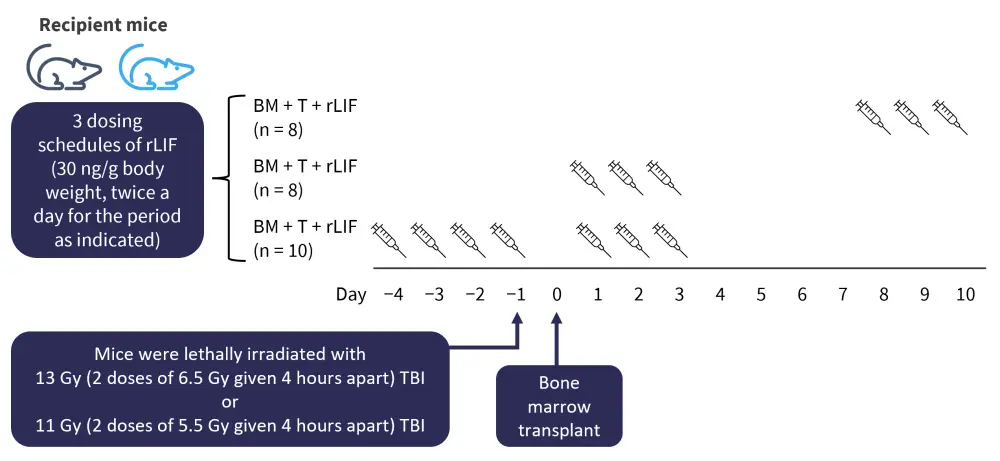
BM, bone marrow; rLIF, recombinant LIF protein; T, T cells; TBI, total body irradiation.
*Adapted from Wang, et al.1
Results
Serum LIF levels
Serum LIF levels were evaluated using enzyme-linked immunosorbent assays. In mice who had received total body irradiation (TBI), syngeneic BMT, or allogeneic bone marrow, serum LIF levels were found to be elevated, with significant elevation 2 days after TBI. However, in mice who received allo-BMT, serum LIF levels were elevated to a much higher level, with prolonged elevation seen to Day 14 after treatment in B6C3F1 and Day 20 in C57BL/6 mice. Elevated serum LIF was positively correlated with survival post-transplant, even in the B6C3F1 mice who developed GvHD after transplant. In addition, in the LIF-knockout mice, there was significantly increased GvHD severity and decreased survival compared to the wild-type recipient mice. Together, these findings suggest that LIF is protective against GvHD severity and lethality.
Donor T-cell activation
TBI prior to BMT can cause development of GvHD. This is due to TBI elevating major histocompatibility complex class II (MHC‑II) levels on intestinal epithelial cells in the intestinal crypt region, leading to higher levels of donor T-cell activation, causing GvHD. This elevation in MHC‑II levels after TBI and allo-BMT was seen in both B6C3F1 mice and C57BL/6 mice in this study. However, when rLIF was given, there was a significant decrease in MHC‑II expression on intestinal epithelial cells after transplant, and thus a decrease in donor T-cell activation.
MHC-haploidentical GvHD model (B6C3F1)
Development of GvHD after allo-BMT occurred quickly in B6C3F1 mice. Symptoms included severe weight loss, skin lesions, and diarrhea, with death occurring at ~30 days after transplant. Administration of rLIF protein, starting 4 days before BMT and continuing to 3 days post-BMT, was found to prolong survival and significantly reduce GvHD scores, compared with placebo mice receiving phosphate buffered saline. When rLIF was administered after GvHD development on Days 8−10 post-BMT, a protective effect was still seen, though this was less than when given before transplant.
B6C3F1 mice undergoing allo-BMT were also given recombinant interleukin-12 (rIL-12), which was associated with an increases infiltration of donor immune cells in the spleen and therefore increased spleen weight, which is an indicator of GvHD. Interestingly, in mice who did not receive rLIF while undergoing allo-BMT, rIL-12 had little effect. This suggests that LIF regulation of IL-12 contributes to the protective effect against GvHD of LIF.
In addition, mRNA levels of the proinflammatory cytokines, TNFα, CXCL1, CCL2, IL‑1a, IL‑1b, and IL‑22, were measured by quantitative real-time PCR and found to be significantly increased in B6C3F1 mice in the small intestine at Day 7 post-BMT. However, in mice where rLIF was administered, this cytokine increase was largely not seen.
MHC mismatched GvHD model (C57BL/6)
Similar to the haploidentical GvHD model, administration of rLIF to mice receiving allo-BMT reduced GvHD severity. In mice receiving the higher TBI dose of 13 Gy, rLIF was shown to give the most significant protection against GvHD-induced injury in the small intestine.
To test the effect of rLIF administration on graft-versus-leukemia activity, some C57BL/6 mice received both BM and C1498 murine acute myeloid leukemia cells. These mice quickly developed leukemia and died by Day 28 post-BM. Use of rLIF in these mice inhibited the development of leukemia and also prolonged survival by an average of 5 days.
Conclusion
The study by Wang et al. demonstrated the protective effect of LIF on GvHD post allo-BMT. Elevated LIF levels were observed in mice after transplant, which was associated with longer survival before succumbing to GvHD. Additional LIF in the form of rLIF also prolonged survival and gave a protective effect against GvHD. rLIF provides this benefit against GvHD at least in part due to decreasing MCH‑II expression in the intestine, leading to decreased donor T-cell activation and prevention of inflammation and damage that indicates GvHD. Use of rLIF did not affect the graft-versus-leukemia activity, suggesting it could be utilized therapeutically as a safe and effective option when undergoing allo-BMT. In both MHC-haploidentical and MHC-mismatched GvHD models, mice responded well to rLIF and protective effects against GvHD were seen.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content
Your opinion matters
What do you consider to be the main potential advantage of mesenchymal stromal cells in the treatment of acute GvHD?

