All content on this site is intended for healthcare professionals only. By acknowledging this message and accessing the information on this website you are confirming that you are a healthcare professional. If you are a patient or carer, please visit Know GvHD.
The GvHD Hub website uses a third-party service provided by Google that dynamically translates web content. Translations are machine generated, so may not be an exact or complete translation, and the GvHD Hub cannot guarantee the accuracy of translated content. The GvHD Hub and its employees will not be liable for any direct, indirect, or consequential damages (even if foreseeable) resulting from use of the Google Translate feature. For further support with Google Translate, visit Google Translate Help.
The GvHD Hub is an independent medical education platform, sponsored by Medac and supported through grants from Sanofi and Therakos. Funders are allowed no direct influence on our content. The levels of sponsorship listed are reflective of the amount of funding given. View funders.
Now you can support HCPs in making informed decisions for their patients
Your contribution helps us continuously deliver expertly curated content to HCPs worldwide. You will also have the opportunity to make a content suggestion for consideration and receive updates on the impact contributions are making to our content.
Find out more
Create an account and access these new features:
Bookmark content to read later
Select your specific areas of interest
View GvHD content recommended for you
REACH3: 3-year final safety and efficacy analysis
Do you know... In the final analysis of the phase III REACH 3 trial of ruxolitinib in patients with chronic graft-versus-host disease, what was the most commonly reported adverse event across both treatment arms, of any grade?
Corticosteroids are the standard first-line treatment choice for patients with chronic graft-versus-host disease (cGvHD). While generally an effective treatment option, many patients prove unresponsive to the treatment, or go on to become refractory or dependent on corticosteroids.1
Ruxolitinib, an inhibitor of the tyrosine kinases Janus kinase (JAK)1 and JAK2, is approved by the U.S. Food and Drug Administration (FDA) for the treatment for cGvHD in patients ≥12 years after failure of one or two lines of systemic therapy. Additionally, ruxolitinib is approved by the European Commission to treat acute and chronic forms of GvHD in patients aged ≥12 years who have inadequate response to corticosteroids.
The phase III REACH3 study (NCT03112603) was designed to evaluate the efficacy and safety of ruxolitinib as a treatment for cGvHD in patients ≥12 years who were unresponsive or refractory to corticosteroid treatment. Primary results were positive, demonstrating the superior efficacy of ruxolitinib vs best available treatment (BAT).1
During the 65th American Society of Hematology (ASH) Annual Meeting and Exposition, Robert Zeiser presented final, long-term efficacy and safety outcomes from the REACH3 trial. Here, we summarize the key results.
Efficacy results (full analysis set)1
- Failure-free survival (FFS) was substantially longer with ruxolitinib than BAT
- Median FFS, 38.4 months vs 5.7 months, hazard ratio (95% CI [confidence interval]): 0.361 (0.268, 0.485)
- 12-month FFS probabilities (95% CI)
- Ruxolitinib: 64.0% (56.1, 70.8)
- BAT: 28.8% (21.8, 36.1)
- FFS probability sustained at 36 months
- Ruxolitinib: 56.5% (48.5, 63.7)
- BAT 18.2% (12.5, 24.9)
- Median duration of response (DOR) was not reached for ruxolitinib vs 6.4 months for BAT
- DOR probability (95% CI) at 12 months
- Ruxolitinib: 70.2% (61.4, 77.5)
- BAT: 39.8% (30.4, 49.1)
- Proportion with DOR (95% CI) sustained at 36 months
- Ruxolitinib: 59.6% (50.4, 67.6)
- BAT: 26.7% (18.5, 35.5)
Median overall survival was not reached in either group, and no statistical difference in risk of death was observed between the treatment groups. Notably, 77% of patients treated with ruxolitinib and 75% of those treated with BAT survived for 3 years.
- Patients who crossed over to ruxolitinib after inadequate response to BAT achieved:
- Overall response rate (95% CI): 50.0% (37.8, 62.2)
- Complete response (CR): 5.7%; partial response (PR): 44.3%
- Best overall response (95% CI): 81.4% (70.3, 89.7)
- CR:7.1%; PR:74.3%
- Overall response rate (95% CI): 50.0% (37.8, 62.2)
Safety results (full analysis set)1
Most patients treated during the main treatment period experienced ≥1 adverse event (AE). Generally, AE rates were higher in patients treated with ruxolitinib vs those treated with BAT (Figure 1), with no new safety signals observed.
AEs leading to treatment discontinuation were reported in 24.2% and 11.% of patients in the ruxolitinib and BAT treatment arms, respectively.
Anemia was the most common any-grade (ruxolitinib: 33.9%, BAT: 15.8%) and Grade ≥3 (ruxolitinib: 17.6%, BAT: 9.5%) AE across all groups. Anemia (24.2%; Grade ≥3, 10.3%) and thrombocytopenia (3.8%) were the most common ruxolitinib and BAT treatment-related AEs, respectively.
Pneumonia was the most common serious AE (all groups) and AE leading to treatment discontinuation (ruxolitinib group only). On-treatment deaths were largely due to cGvHD (ruxolitinib: 55.6%, BAT 50.0%).
Figure 1. Overview of safety (Grade ≥3 events) in the ruxolitinib and BAT treatment arms
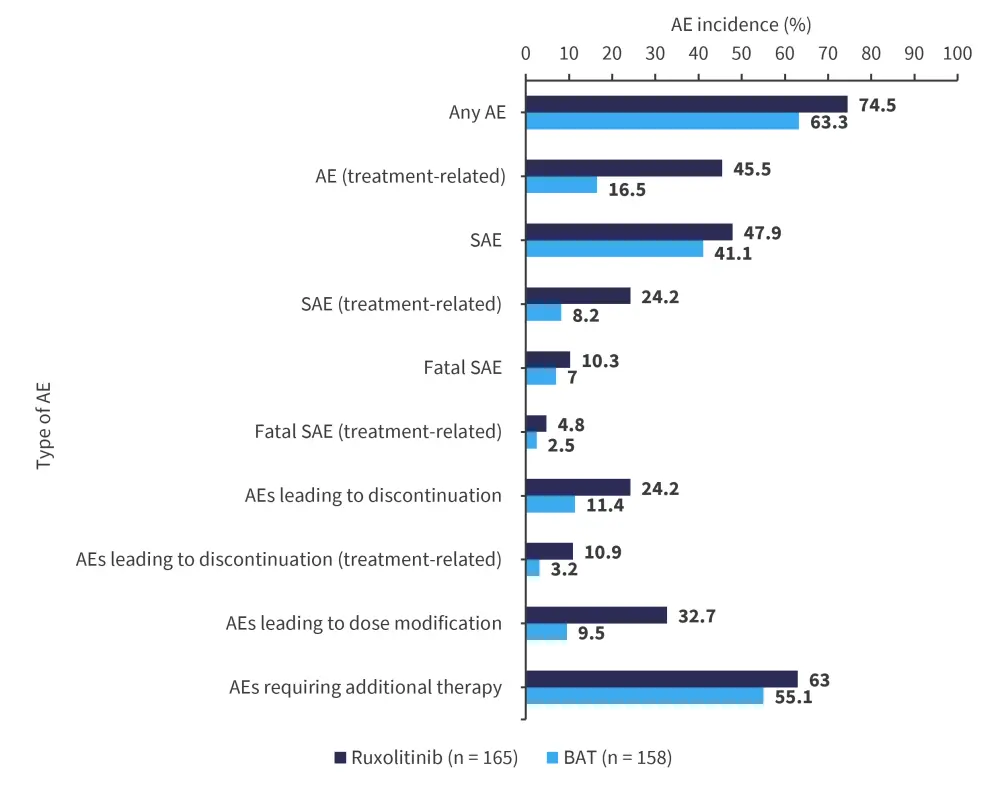
AE, adverse event; BAT, best available treatment; SAE, serious adverse event.
*Data from Zeiser.1
Conclusion
After 3 years of treatment or follow-up in REACH3, ruxolitinib showed longer FFS and higher DOR compared with BAT, indicating that cGvHD was more manageable with ruxolitinib treatment. Patients who crossed over to ruxolitinib from BAT were still able to achieve high response rates, in line with patients randomized to ruxolitinib. Ruxolitinib was shown to be well tolerated with no new safety signals recorded. Overall, the long-term control of cGvHD and tolerability of ruxolitinib was demonstrated in this patient group.
References
Please indicate your level of agreement with the following statements:
The content was clear and easy to understand
The content addressed the learning objectives
The content was relevant to my practice
I will change my clinical practice as a result of this content




